 13
13 The correct answer is D) “world desctruction is inevitable if the United States and the Soviet Union continue to compete fot military superiority.
The response that best describes Kennedy’s argument in the paragraph is “world desctruction is inevitable if the United States and the Soviet Union continue to compete fot military superiority.”
This is part of the speech John F. Kennedy made in his Inaugural Address. In his argument, he states that the United States and the Soviet Union must work together to avoid nuclear work. With the use of his well though words, Kennedy is trying to forecast the worst scenario for the people to see. He says “neither two of this powerful nations can take comfort from its present course; both sides overburn by the cost of modern weapons, both rightly alarmed by the spread of the modern atom, yet both racing to alter that uncertain balance of terror that stays the hand of mankind’s final word.
Kennedy was trying to create consciousness about the consequences of another war if the race of arms continued the way it was at that moment.
 13
13 The correct answer is D) “world desctruction is inevitable if the United States and the Soviet Union continue to compete fot military superiority.
The response that best describes Kennedy’s argument in the paragraph is “world desctruction is inevitable if the United States and the Soviet Union continue to compete fot military superiority.”
This is part of the speech John F. Kennedy made in his Inaugural Address. In his argument, he states that the United States and the Soviet Union must work together to avoid nuclear work. With the use of his well though words, Kennedy is trying to forecast the worst scenario for the people to see. He says “neither two of this powerful nations can take comfort from its present course; both sides overburn by the cost of modern weapons, both rightly alarmed by the spread of the modern atom, yet both racing to alter that uncertain balance of terror that stays the hand of mankind’s final word.
Kennedy was trying to create consciousness about the consequences of another war if the race of arms continued the way it was at that moment.
assessments
Explanation:
Assessment refers to the technique that is used to make the interference for the students so that they can learned and developed. It includes various things like designing, collecting, analyzing, interpreting, etc
Now for the technology, the evaluation technique is required so that the benefits and risk can be analyzed also the best action to be carried out for coming up with the positive results
So the given situation represents the assessments
assessments
Explanation:
Assessment refers to the technique that is used to make the interference for the students so that they can learned and developed. It includes various things like designing, collecting, analyzing, interpreting, etc
Now for the technology, the evaluation technique is required so that the benefits and risk can be analyzed also the best action to be carried out for coming up with the positive results
So the given situation represents the assessments
Answer:
AStep-by-step explanation:
The input force is 50 N. But it will not create not any change. No mechanical advantage is observed.

Answer:
Taking into accoun the ideal gas law, The volume of a container that contains 24.0 grams of N2 gas at 328K and 0.884 atm is 26.07 L.
An ideal gas is a theoretical gas that is considered to be composed of point particles that move randomly and do not interact with each other. Gases in general are ideal when they are at high temperatures and low pressures.
The pressure, P, the temperature, T, and the volume, V, of an ideal gas, are related by a simple formula called the ideal gas law:
P×V = n×R×T
where P is the gas pressure, V is the volume that occupies, T is its temperature, R is the ideal gas constant, and n is the number of moles of the gas. The universal constant of ideal gases R has the same value for all gaseous substances.
Explanation:
In this case, you know:
P= 0.884 atm
V= ?
n= 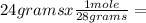 0.857 moles (where 28 g/mole is the molar mass of N₂, that is, the amount of mass that the substance contains in one mole.)
0.857 moles (where 28 g/mole is the molar mass of N₂, that is, the amount of mass that the substance contains in one mole.)
R=0.082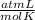
T= 328 K
Replacing in the ideal gas law:
0.884 atm×V= 0.857 moles× 0.082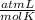 ×328 K
×328 K
Solving:
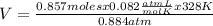
V= 26.07 L
The volume of a container that contains 24.0 grams of N2 gas at 328K and 0.884 atm is 26.07 L.

It will provide an instant answer!
