D. Point into Leaf: water(H₂O), carbon dioxide (CO₂) sunlight, Point away from leaf: Sugars, oxygen (O₂)
Explanation:
Photosynthesis is the process used in the production of chemical energy in the form of energy containing organic compounds and oxygen from atmospheric carbon dioxide and liquid water enabled with the energy captured from light (mainly Sunlight) by the chlorophyll in green plants and other organisms
The basic chemical reaction involved in a photosynthesis reaction is presented as follows;
 Sunlight
Sunlight
 ↓
↓
 CO₂ + H₂O (l) → C₆H₁₂O₆ + O₂
CO₂ + H₂O (l) → C₆H₁₂O₆ + O₂
Carbon dioxide  Water Glucose Oxygen
Water Glucose Oxygen
D. Point into Leaf: water(H₂O), carbon dioxide (CO₂) sunlight, Point away from leaf: Sugars, oxygen (O₂)
Explanation:
Photosynthesis is the process used in the production of chemical energy in the form of energy containing organic compounds and oxygen from atmospheric carbon dioxide and liquid water enabled with the energy captured from light (mainly Sunlight) by the chlorophyll in green plants and other organisms
The basic chemical reaction involved in a photosynthesis reaction is presented as follows;
 Sunlight
Sunlight
 ↓
↓
 CO₂ + H₂O (l) → C₆H₁₂O₆ + O₂
CO₂ + H₂O (l) → C₆H₁₂O₆ + O₂
Carbon dioxide  Water Glucose Oxygen
Water Glucose Oxygen
 18
18 1. It can be done alone or with a friend.
2. the ability to maintain stable levels of oxygen and blood flow during an extended period of physical activity
3. Be inactive.
4. Type I diabetes
5. community
6. tai chi
7. lack of confidence
8. baseball
9. 5 to 10 minutes
10. efficacy
11. rhythm and sequencing
12. Is it scientifically based?
13. the ability of muscles to function over a period of time without becoming tired
14. stating when and how often you will do the steps
15. Increase endurance over a limited amount of time.
16. scammer
17. a mix of low, moderate, and vigorous
18. intensity
19. 15
20. Pilates
 18
18 1. It can be done alone or with a friend.
2. the ability to maintain stable levels of oxygen and blood flow during an extended period of physical activity
3. Be inactive.
4. Type I diabetes
5. community
6. tai chi
7. lack of confidence
8. baseball
9. 5 to 10 minutes
10. efficacy
11. rhythm and sequencing
12. Is it scientifically based?
13. the ability of muscles to function over a period of time without becoming tired
14. stating when and how often you will do the steps
15. Increase endurance over a limited amount of time.
16. scammer
17. a mix of low, moderate, and vigorous
18. intensity
19. 15
20. Pilates
Answer:
AStep-by-step explanation:
The input force is 50 N. But it will not create not any change. No mechanical advantage is observed.
 1
1 Answer:
52.6 gramStep-by-step explanation:
It is clear by the equation 2(27+3×35.5)= 267 gm of AlCl3 reacts with 6× 80 = 480 gm of Br2 . So 29.2 gm reacts = 480× 29.2/267= 52.6 gm
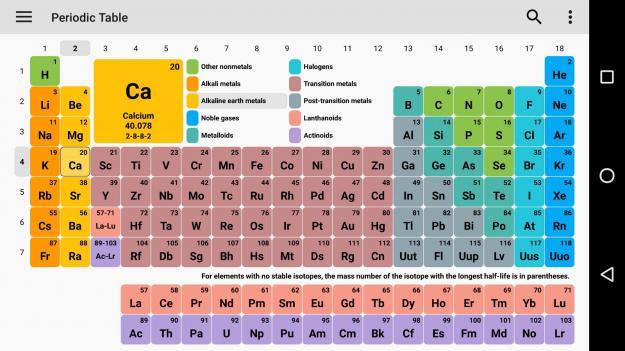
Calcium (Ca)(On the periodic table, ionization energy increases as you go up and to the right of the periodic table)
glycoproteins
Explanation:
A positive reaction for Molisch's test is given by almost all carbohydrates (exceptions include tetroses & trioses). It can be noted that even some glycoproteins and nucleic acids give positive results for this test (since they tend to undergo hydrolysis when exposed to strong mineral acids and form monosaccharides).
Answer:
Taking into accoun the ideal gas law, The volume of a container that contains 24.0 grams of N2 gas at 328K and 0.884 atm is 26.07 L.
An ideal gas is a theoretical gas that is considered to be composed of point particles that move randomly and do not interact with each other. Gases in general are ideal when they are at high temperatures and low pressures.
The pressure, P, the temperature, T, and the volume, V, of an ideal gas, are related by a simple formula called the ideal gas law:
P×V = n×R×T
where P is the gas pressure, V is the volume that occupies, T is its temperature, R is the ideal gas constant, and n is the number of moles of the gas. The universal constant of ideal gases R has the same value for all gaseous substances.
Explanation:
In this case, you know:
P= 0.884 atm
V= ?
n= 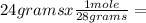 0.857 moles (where 28 g/mole is the molar mass of N₂, that is, the amount of mass that the substance contains in one mole.)
0.857 moles (where 28 g/mole is the molar mass of N₂, that is, the amount of mass that the substance contains in one mole.)
R=0.082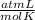
T= 328 K
Replacing in the ideal gas law:
0.884 atm×V= 0.857 moles× 0.082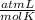 ×328 K
×328 K
Solving:
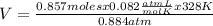
V= 26.07 L
The volume of a container that contains 24.0 grams of N2 gas at 328K and 0.884 atm is 26.07 L.

It will provide an instant answer!
