 36
36 The mistake is when you book them together the molecules have different energy and the system is not in equilibrium.
Given data:
Jack's model consists of 1 object = 2 molecules.
And, object 2 = 6 molecules.
And there were 16 energy cubes kept in each object.
As per the given problem, the model developed by Jack has the same energy for each of the two objects, but as each object is made up of a different number of molecules, in the system with more molecules, object 2, each one has approximately 2.4 less energy and the molecules of Object 1 have an E / 2 energy.
Thus, we can conclude that the mistake is when you book them together the molecules have different energy and the system is not in equilibrium.
learn more about the thermal equilibrium here:
link

 1
1 Answer:
52.6 gramStep-by-step explanation:
It is clear by the equation 2(27+3×35.5)= 267 gm of AlCl3 reacts with 6× 80 = 480 gm of Br2 . So 29.2 gm reacts = 480× 29.2/267= 52.6 gm
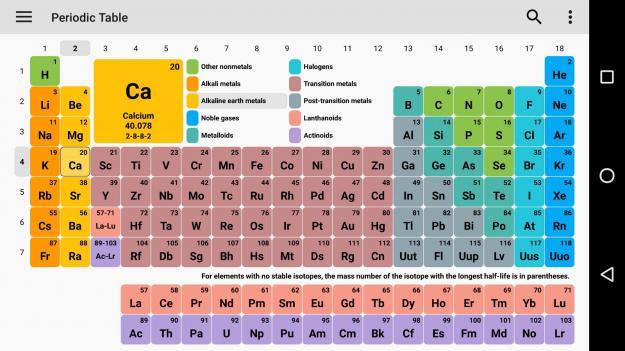
Calcium (Ca)(On the periodic table, ionization energy increases as you go up and to the right of the periodic table)
 1
1 
It will provide an instant answer!
