 3
3 2Fe + O₂ > 2FeO
8 mol Fe produce
8 mol Fe * 2 mol FeO / 2 mol Fe = 8 mol FeO
Mass of FeO = 8 mol FeO * 71.85 g/mol = 574.8 grams FeO
Explanation:
Having 8 mol of Iron means 8 moles of iron oxide can be produced. Each mole of iron oxide has a molecular weight of 71.85 grams. Therefore, 8 moles of iron oxide should weight 574.8 grams.
The answer to your question is 576 grams of FeO
Explanation:
Balanced chemical reaction
2Fe + O₂ ⇒ 2 FeO
Reactant Elements Products
2 Fe 2
2 O 2
Data
mass of FeO = ?
moles of Fe = 8
Excess Oxygen
Process
1.- Convert moles of Fe to grams
Atomic mass = 56 g
56 g of Fe 1 mol
x 8 moles
x = (8 x 56) / 1
x = 448 g of Fe
2.- Use the coefficients of the balanced reaction to find the grams of FeO.
Molecular mass of FeO = 2[ 56 + 16]
= 144 g
2(56) g of Fe 144 g of FeO
448 g x
x = (448 x 144) / 2(56)
x = 64512 / 112
x = 576 grams of FeO
 2
2 Answer : Mass of copper (Cu) metal obtained = 556.281 g
Solution : Given,
Mass of copper ore = 1 Kg = 1000 g
From the periodic table, the molar mass of copper and copper ore are
Molar mass of copper = 63.546 g/mole
Molar mass of copper ore, 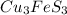 = 342.678 g/mole
= 342.678 g/mole
First we have to calculate the number of moles of copper ore, 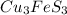
Moles of 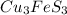 =
= 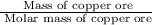 =
= 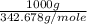 = 2.918 moles
= 2.918 moles
The balanced chemical reaction is :
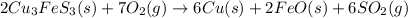
From this chemical reaction, we conclude that
2 moles of 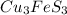 gives → 6 moles of Cu
gives → 6 moles of Cu
2.918 moles of 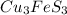 gives →
gives → 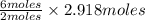
= 8.754 moles
Thus, the Moles of copper (Cu) = 8.754 moles
Now, calculate the Mass of copper (Cu).
Mass of copper (Cu) = Moles of copper (Cu) × Molar mass of copper (Cu)
= 8.757 moles × 63.546 g/moles
= 556.281 g
 2
2 Answer : Mass of copper (Cu) metal obtained = 556.281 g
Solution : Given,
Mass of copper ore = 1 Kg = 1000 g
From the periodic table, the molar mass of copper and copper ore are
Molar mass of copper = 63.546 g/mole
Molar mass of copper ore, 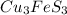 = 342.678 g/mole
= 342.678 g/mole
First we have to calculate the number of moles of copper ore, 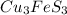
Moles of 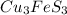 =
= 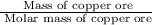 =
= 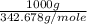 = 2.918 moles
= 2.918 moles
The balanced chemical reaction is :
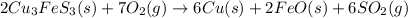
From this chemical reaction, we conclude that
2 moles of 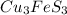 gives → 6 moles of Cu
gives → 6 moles of Cu
2.918 moles of 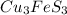 gives →
gives → 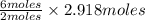
= 8.754 moles
Thus, the Moles of copper (Cu) = 8.754 moles
Now, calculate the Mass of copper (Cu).
Mass of copper (Cu) = Moles of copper (Cu) × Molar mass of copper (Cu)
= 8.757 moles × 63.546 g/moles
= 556.281 g

Answer:
AStep-by-step explanation:
The input force is 50 N. But it will not create not any change. No mechanical advantage is observed.
 1
1 Answer:
52.6 gramStep-by-step explanation:
It is clear by the equation 2(27+3×35.5)= 267 gm of AlCl3 reacts with 6× 80 = 480 gm of Br2 . So 29.2 gm reacts = 480× 29.2/267= 52.6 gm
Calcium (Ca)(On the periodic table, ionization energy increases as you go up and to the right of the periodic table)
glycoproteins
Explanation:
A positive reaction for Molisch's test is given by almost all carbohydrates (exceptions include tetroses & trioses). It can be noted that even some glycoproteins and nucleic acids give positive results for this test (since they tend to undergo hydrolysis when exposed to strong mineral acids and form monosaccharides).

It will provide an instant answer!
