Here's what I get
Explanation:
(g) Titration curves
I can't draw two curves on the same graph, but I can draw two separate curves for you.
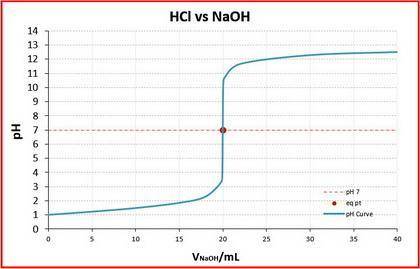
The graph in part (d) had an equivalence point at 20 mL.
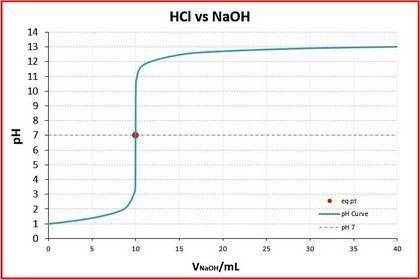
In the second titration, the NaOH was twice as concentrated, so the volume to equivalence point would be half as much — 10 mL.
The two titration curves are below.
(h) Evidence of reaction
HCl and NaOH are both colourless.
They don't evolve a gas or form a precipitate when they react.
The student probably noticed that the Erlenmeyer flask warmed up — a sign of a chemical change.