 3
3 Knowing that Quindlen wrote the text in response to the attacks of September 11, I better understand her viewpoint that tragedy often unites American citizens. In addition, I understand how Quindlen wants to persuade readers to rethink the meaning and value of American identity.
Explanation:

 46
46 (i) Americans are united at time of hardship.
(ii) It concentrates on the diversity of the American society and their unity.
Quindlen's piece was written in context of the aftermath of the terrorist attack of 9/11. It concentrated on how the Americans overcame the trauma.
 4
4 The arguments provided can be determining to follow the author's posture however they can be contrasted so I can't really accept it and follow the author's Idea. They are not as strong to do so, at least for me.
Explanation:
First of all, I have to say that to understand the central Idea I have to identify the main topic. We have the crisis, the consequences, and origin. But, all of those come together to construct the main topic, they speak and describe the same element to provide principal and secondary ideas about it. Second, the posture that the author took is very obvious, in every text, the author will state comments in favor or opposing the object of study and provide arguments with enough evidence to support his or her claims. Then, in third place after analyzing the whole posture and object I just needed to establish a connection between both and see if they convinced me to accept his point of view as factually correct or not. They had enough evidence, but I reserve my position as arbitrary because I believe it wasn’t that strong to perceive it as the only truth.
 4
4 The arguments provided can be determining to follow the author's posture however they can be contrasted so I can't really accept it and follow the author's Idea. They are not as strong to do so, at least for me.
Explanation:
First of all, I have to say that to understand the central Idea I have to identify the main topic. We have the crisis, the consequences, and origin. But, all of those come together to construct the main topic, they speak and describe the same element to provide principal and secondary ideas about it. Second, the posture that the author took is very obvious, in every text, the author will state comments in favor or opposing the object of study and provide arguments with enough evidence to support his or her claims. Then, in third place after analyzing the whole posture and object I just needed to establish a connection between both and see if they convinced me to accept his point of view as factually correct or not. They had enough evidence, but I reserve my position as arbitrary because I believe it wasn’t that strong to perceive it as the only truth.


 1
1 Answer:
52.6 gramStep-by-step explanation:
It is clear by the equation 2(27+3×35.5)= 267 gm of AlCl3 reacts with 6× 80 = 480 gm of Br2 . So 29.2 gm reacts = 480× 29.2/267= 52.6 gm
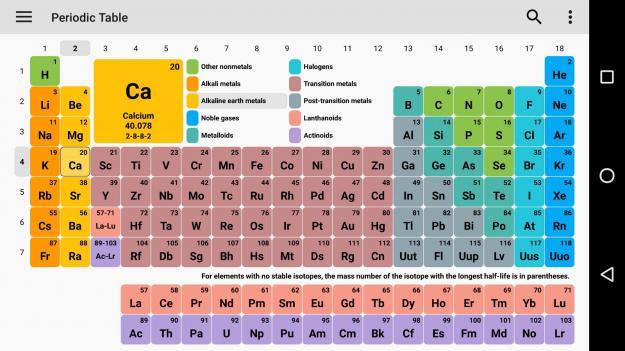
Calcium (Ca)(On the periodic table, ionization energy increases as you go up and to the right of the periodic table)
glycoproteins
Explanation:
A positive reaction for Molisch's test is given by almost all carbohydrates (exceptions include tetroses & trioses). It can be noted that even some glycoproteins and nucleic acids give positive results for this test (since they tend to undergo hydrolysis when exposed to strong mineral acids and form monosaccharides).
Answer:
Taking into accoun the ideal gas law, The volume of a container that contains 24.0 grams of N2 gas at 328K and 0.884 atm is 26.07 L.
An ideal gas is a theoretical gas that is considered to be composed of point particles that move randomly and do not interact with each other. Gases in general are ideal when they are at high temperatures and low pressures.
The pressure, P, the temperature, T, and the volume, V, of an ideal gas, are related by a simple formula called the ideal gas law:
P×V = n×R×T
where P is the gas pressure, V is the volume that occupies, T is its temperature, R is the ideal gas constant, and n is the number of moles of the gas. The universal constant of ideal gases R has the same value for all gaseous substances.
Explanation:
In this case, you know:
P= 0.884 atm
V= ?
n= 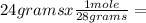 0.857 moles (where 28 g/mole is the molar mass of N₂, that is, the amount of mass that the substance contains in one mole.)
0.857 moles (where 28 g/mole is the molar mass of N₂, that is, the amount of mass that the substance contains in one mole.)
R=0.082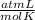
T= 328 K
Replacing in the ideal gas law:
0.884 atm×V= 0.857 moles× 0.082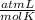 ×328 K
×328 K
Solving:
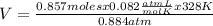
V= 26.07 L
The volume of a container that contains 24.0 grams of N2 gas at 328K and 0.884 atm is 26.07 L.

It will provide an instant answer!
