 1
1 If a runner exerts 350 J of work to generate 175 W of power, it took the runner 2 seconds to do the work.
Explanation:
Power is defined as the speed with which a job is done. In other words, power is a given amount of work done in a given unit of time. That is, it is the amount of work per unit of time that some object or system produces.
Its expression is:
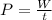
where:
P: Power developed by the force doing the work. Its unit of measure in the International System is the Watt (W) W: Work. Its unit of measure in the International System is the Joule (J) t: Time during which the work is carried out. Its unit of measurement in the International System is the second (s).In this case:
P= 175 WW= 350 Jt=?Replacing:
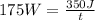
and solving you get:
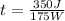
t= 2 s
If a runner exerts 350 J of work to generate 175 W of power, it took the runner 2 seconds to do the work.
 1
1 If a runner exerts 350 J of work to generate 175 W of power, it took the runner 2 seconds to do the work.
Explanation:
Power is defined as the speed with which a job is done. In other words, power is a given amount of work done in a given unit of time. That is, it is the amount of work per unit of time that some object or system produces.
Its expression is:
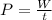
where:
P: Power developed by the force doing the work. Its unit of measure in the International System is the Watt (W) W: Work. Its unit of measure in the International System is the Joule (J) t: Time during which the work is carried out. Its unit of measurement in the International System is the second (s).In this case:
P= 175 WW= 350 Jt=?Replacing:
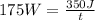
and solving you get:
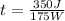
t= 2 s
If a runner exerts 350 J of work to generate 175 W of power, it took the runner 2 seconds to do the work.
 11
11 Explanation:
p=w/t
t=w/p
=350/125
t=2.8s

Calcium (Ca)(On the periodic table, ionization energy increases as you go up and to the right of the periodic table)
glycoproteins
Explanation:
A positive reaction for Molisch's test is given by almost all carbohydrates (exceptions include tetroses & trioses). It can be noted that even some glycoproteins and nucleic acids give positive results for this test (since they tend to undergo hydrolysis when exposed to strong mineral acids and form monosaccharides).
Answer:
Taking into accoun the ideal gas law, The volume of a container that contains 24.0 grams of N2 gas at 328K and 0.884 atm is 26.07 L.
An ideal gas is a theoretical gas that is considered to be composed of point particles that move randomly and do not interact with each other. Gases in general are ideal when they are at high temperatures and low pressures.
The pressure, P, the temperature, T, and the volume, V, of an ideal gas, are related by a simple formula called the ideal gas law:
P×V = n×R×T
where P is the gas pressure, V is the volume that occupies, T is its temperature, R is the ideal gas constant, and n is the number of moles of the gas. The universal constant of ideal gases R has the same value for all gaseous substances.
Explanation:
In this case, you know:
P= 0.884 atm
V= ?
n= 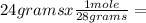 0.857 moles (where 28 g/mole is the molar mass of N₂, that is, the amount of mass that the substance contains in one mole.)
0.857 moles (where 28 g/mole is the molar mass of N₂, that is, the amount of mass that the substance contains in one mole.)
R=0.082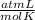
T= 328 K
Replacing in the ideal gas law:
0.884 atm×V= 0.857 moles× 0.082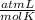 ×328 K
×328 K
Solving:
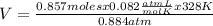
V= 26.07 L
The volume of a container that contains 24.0 grams of N2 gas at 328K and 0.884 atm is 26.07 L.

It will provide an instant answer!
