1. Protons and neutrons have the same charge.
Protons have positive charge, equal to 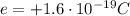 , while neutrons have zero charge.
, while neutrons have zero charge.
2. mass number
The mass number of an atom is equal to the sum of protons and neutrons inside its nucleus.
3. Atoms are made up of smaller particles.
According to Dalton's theory, atoms are the smallest particles that make matter, and they are indivisible and indestructible, so they are NOT made up of smaller particles.
4. a solid sphere
In Dalton's theory, atoms are not made of smaller particles, so we can think them as solid spheres.
5. J. J. Thomson
In his experiment with cathode ray tubes, JJ Thomson demonstrated the existance of the electrons, which are negatively charged particles inside the atom. In his model of the atom (plum-pudding model), Thomson thought the atom consists of a uniform positive charge and the electrons are located inside this positive charge.
6. An electron has the same amount of energy in all orbitals.
In fact, each orbital corresponds to a different energy level: the farther the orbital from the nucleus, the higher the energy of the electrons contained in that orbital.
7. A hydrogen atom in heavy water has an extra neutron.
Heavy water is a type of water that contains deuterium, which is an isotope of the hydrogen consisting of one proton and one neutron (so, one extra neutron).
8. The glowing beam was always deflected by charged plates
In his cathode's ray tube experiment, Thomson shows that the beam of unknown particles (= the electrons) were deflected by charge plates, so the particles had to be also electrically charged.
9. electrons move to a lower energy level
When electrons move from a higher energy level to a lower energy, they emit a photon (light) of energy equal to the difference in energy between the two energy levels.
10. orbital
In quantum mechanics, electrons in the atom are not precisely located, since we cannot determine their exact position and velocity at the same time. Therefore, we can only describe regions of space where the electrons have a certain probability to be found, and these regions of space are called orbitals.
11. 14
According to Dalton's theory, the proportions of the reactants must be respected in order to form the same compound. Therefore, we can write:
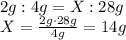
12. negative charge, found outside the nucleus
Electrons are particles with negative charge of magnitude 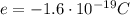 that orbit around the nucleus. The nucleus, instead, consists of protons (positively charged, with charge opposite to the electron) and neutrons (neutrally charged).
that orbit around the nucleus. The nucleus, instead, consists of protons (positively charged, with charge opposite to the electron) and neutrons (neutrally charged).
13. move from higher to lower energy levels
When electrons move from a higher energy level to a lower energy inside a neon atom, they emit a photon (which is light) whose energy is equal to the difference in energy between the two energy levels.
14. atomic number from its mass number
In fact:
- the atomic number of an atom (Z) is equal to the number of protons inside the nucleus
- the mass number of an atom (A) is equal to the sum of protons+neutrons inside the nucleus
Therefore, we can find the number of neutrons in the nucleus by calculating the difference between A and Z:
Number of neutrons = A - Z
15. None of them
None of these examples is a good analogy to describe the location of an electron in an atomic orbital: in fact, the position of an electron in an orbital cannot be precisely described, we can only describe the probability to find the electron in a certain position, and none of these example is an analogy of this model.