 36
36 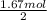
 161
161 1) 0.835 mol O2
2) 1.47 mol 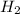
Explanation:
just did it on edu
 36
36 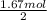
 14
14  14
14  5
5  13
13 13.7 moles of O₂ are needed
Explanation:
In order to find the moles of reactants that may react to make the products we need to determine the reaction:
Reactants are hydrogen and oxygen
Product: Water
2 moles of hydrogen can react to 1 mol of oxygen and produce 2 moles of water.
Balanced reaction: 2H₂(g) + O₂(g) → 2H₂O(l)
If 2 moles of hydrogen need 1 mol of oxygen to react
Therefore, 27.4 moles of H₂ must need (27.4 .1) / 2 = 13.7 moles of O₂
 74
74  1
1 13.7 moles of oxygen (O₂) are required to completely react with 27.4 mol of H₂.
Explanation:
The complete question is:
"The following balanced equation shows the formation of water.
2H₂ + O₂ ----> 2H₂O
How many moles of oxygen (O₂) are required to completely react with 27.4 mol of H₂?
A: 6.8 mol
B: 13.7 mol
C: 54.8 mol
D: 109.6 mol"
The rule of three or is a way of solving problems of proportionality between three known values and an unknown value, establishing a relationship of proportionality between all of them. That is, what is intended with it is to find the fourth term of a proportion knowing the other three. Remember that proportionality is a constant relationship or ratio between different magnitudes.
If the relationship between the magnitudes is direct, that is, when one magnitude increases, so does the other (or when one magnitude decreases, so does the other), the direct rule of three must be applied. To solve a direct rule of three, the following formula must be followed:
a ⇒ b
c ⇒ x
So:
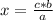
The rule of three can be applied in this case in the following way: by stoichiometry of the reaction (that is, the relationship between the amount of reagents and products in a chemical reaction), you know that 2 moles of H₂ react 1 mole of O₂. If there are 27.4 moles of H₂, how many moles of O₂ will react?
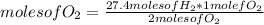
moles of O₂=13.7
13.7 moles of oxygen (O₂) are required to completely react with 27.4 mol of H₂.
 12
12 
It will provide an instant answer!
